アルツハイマー病を遅らせる新薬
ある医師が治験として、ある薬を患者に飲ませところ、一晩だけ認知症が改善し、みんなでダンスを踊ることができたという映画を昔見たことがあります。いつもタイトルが思い出せずにいます。(軽い認知症かも?) 誰か知りませんか?
VOAで英語を学びましょう!!
アルツハイマー病の進行を遅らせる新薬、しかし治療薬ではない(和訳)
New Drug Slows Alzheimer's, But Not a Cure
Jan.10,2022
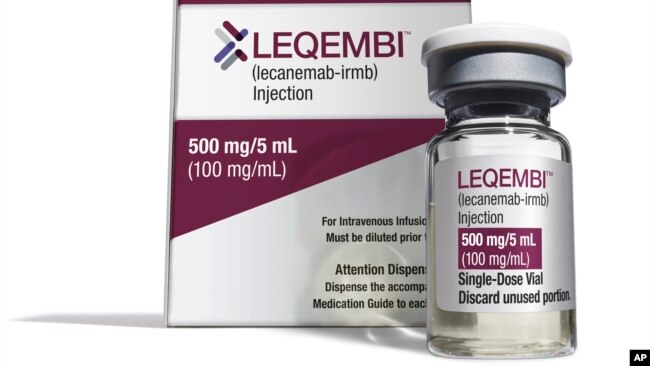
米国の保健当局は先週、新しいアルツハイマー病治療薬を承認しました。Leqembi(レケンビ)と呼ばれるこの薬は、記憶の衰えを数ヶ月遅らせる初めてのものです。
アルツハイマー病協会では、アルツハイマー病を、記憶力、思考力、推理力の低下を引き起こす脳の病気と説明しています。
アルツハイマー病の最も一般的な兆候の1つは、最近学んだ情報や日付、出来事を忘れることです。以前はできていたことが困難になります。議論に参加したり、ついていくことが苦手な人もいます。
今回承認されたLeqembi(レケンビ)は、この病気に対して長い間必要とされていた新しい処置です。
しかし、専門家は、この薬は治療薬ではないと警告しています。
ジョイ・スナイダー博士は、ミズーリ州セントルイスにあるワシントン大学の脳神経外科医です。彼女は、「この薬は治療薬ではありません。悪化するのを止めることはできませんが、病気の進行はかなり遅らせることができます 」と言っています。彼女はさらに、「それは、運転できる期間が6ヶ月から1年延びることを意味するかもしれません 」と付け加えています。
米国食品医薬品局(FDA)は、通常より迅速なプロセスでこの薬を承認しました。このプロセスでは、早期の結果に基づいて医薬品を発売することが認められています。
この薬は早期の患者のみを対象とし、2週間ごとに医療従事者による注射が必要です。どの程度、この病気の患者さんに役立つかは、まだはっきりしていません。また、安全性についても懸念されています。
それでも、「これはlandmark画期的なことです」と国立老化研究所のリチャード・ホデス所長は言います。「十分とは言えませんが、何かできることがあるというのはencouraging 励みになります」と付け加えます。
新薬はどのように作用するのか?
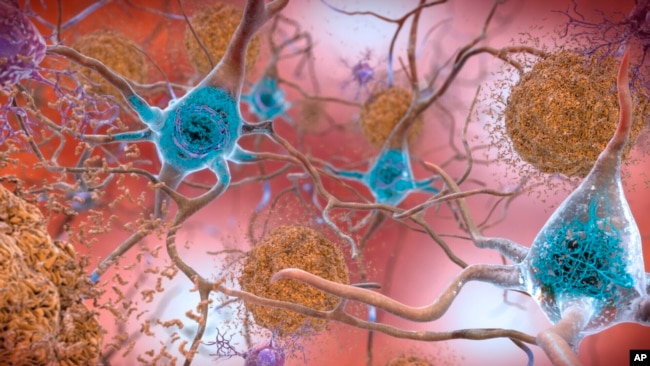
Leqembi(レケンビ)は、日本のエーザイと米国のパートナーであるバイオジェンによって製造されています。この薬は、脳に蓄積してアルツハイマー病を引き起こす物質であるベータアミロイドを標的にして除去するように設計されています。
サム・ガンディ 博士はニューヨークのマウントサイナイ病院のアルツハイマー病の専門家です。彼はAP通信に対して、Leqembi(レケンビ)はこの病気を治療しようとする他の薬とは少し異なった形のアミロイドをターゲットにしていると語っています。このことが、他の多くの実験的医薬品が失敗している中で、Leqembi(レケンビ)が成功した理由を説明しているかもしれません。
2021年、米国の保健当局は、アドヘルムという名の同様の医薬品を承認しましたが、それが本当に患者に役立つとは研究で証明されていません。この動きは、議会の調査の後、批判につながりました。
Leqembi(レケンビ)はどの程度効果があるのか?
1,800人近くを対象としたエーザイの18カ月間の研究では、Leqembi(レケンビ)は早期ステージの患者が悪化するのを5カ月ほど遅らせると思われます。
この研究では、患者の精神的・機能的能力を(18-point scale )18段階で評価しました。この薬を投与された人々は、それでもなお悪化しましたが、不活性物質を投与された人々ほどは早く悪化しませんでした。その差は研究終了時には0.5ポイント近くになっていました。
専門家の間でもこの差にどの程度の意味があるかは意見が分かれています。
マシュー・シュラグ博士はテネシー州のバンダービルト大学の神経学研究者です。彼は、「ほとんどの患者は、その違いに気づかないだろう 」と言っています。
意味のある改善と思っているシュラグ博士や他の研究者は、18点満点で少なくとも1点の差が必要だと考えています。
また、早期 -まだ機能が十分な時 -に病気の進行を遅らせることが重要だと言う人もいます。
国立老化研究所のホデス所長は、「(cognition)認知の数カ月の改善、それはあなたにどんな価値があるのか?」と言います。
さらに、「そこで、強い主張ができると思います:もし、私が家族と(interact) 交流でき、数ヶ月間自立することができれば... それは非常に意味のある(outcome)結果です。」と彼は言います。
リスクは何か?
他のアミロイドを標的とする薬剤と同様に、Leqembi(レケンビ)は脳の腫れや小さな脳出血を引き起こす可能性があります。エーザイの研究では、薬剤投与者の13%に腫脹が、17%に小脳出血がみられました。
その理由として考えられるのは、amyloid plaques アミロイド斑は通常、脳の神経細胞の周りに形成されますが、時には血管の中にも入り込むためです。
Leqembi(レケンビ)使用者の何人かは服用中に死亡しており、その中には血液をサラサラにする薬を服用していた2人が含まれています。エーザイは、アルツハイマー病治療薬は彼らの死亡の原因にはならなかったと発表しています。
しかし、アルツハイマー病の専門家であるガンディ氏は、重篤な出血の危険性が最も高いのは、血液希釈剤を服用しているLeqembi(レケンビ)使用者だろうと言います。高齢者は、脳卒中の予防や治療のために血液希釈剤を服用するのが一般的です。
患者はまた、高体温、胃の不調、血圧の変化を含む可能性のある薬物による反応を経験することがあります。
エーザイは、この薬剤は1月23日までに入手できるはずだと言っています。1年分の治療費は26,500ドルとなる見込みです。しかし、保険会社がこの薬をカバーすれば、ほとんどの人はそれほどの金額を支払う必要はないでしょう。
New Drug Slows Alzheimer's, But Not a Cure
United States health officials last week approved a new Alzheimer’s drug. The drug, called Leqembi, is the first to slow the decline in memory by several months.
The Alzheimer’s Association describes Alzheimer's as a brain disease that causes a decline in memory, thinking and reasoning skills.
One of the most common signs of Alzheimer’s disease is forgetting recently learned information or dates and events. People with the disease have difficulty doing things that they did before. Some may struggle with joining or following a discussion.
The newly approved Leqembi is a long-needed new treatment for the disease.
But experts warn that the drug is not a cure.
Dr. Joy Snider is a brain doctor at Washington University in St. Louis, Missouri. She said, “This drug is not a cure. It doesn’t stop people from getting worse, but it does measurably slow the progression of the disease.” She added, “That might mean someone could have an extra six months to a year of being able to drive.”
The U.S. Food and Drug Administration (FDA) approved the drug through a speedier-than-usual process. The process permits drugs to be launched based on early results.
The drug is only for early-stage patients and requires an injection by healthcare workers every two weeks. It is not yet clear how much the drug can help people with the disease. There are also concerns about its safety.
Still, “it’s a landmark,” said Dr. Richard Hodes, director of the National Institute on Aging. “It’s not enough, but it’s encouraging that there’s something we can do,” he added.
How does the new drug work?
Leqembi is made by Japan’s Eisai and its U.S. partner Biogen. The drug is designed to target and clear away beta-amyloid, a substance that builds up in the brain and leads to Alzheimer’s.
Dr. Sam Gandy is an Alzheimer’s expert at New York’s Mount Sinai Hospital. He told the Associated Press that Leqembi targets a slightly different form of amyloid than other drugs that have sought to treat the disease. That might explain why Leqembi proved to be successful while many other experimental drugs have failed.
In 2021, U.S. health officials approved a similar drug named Aduhelm, even though studies never proved it really helped patients. The move led to criticism after a congressional investigation.
How effective is Leqembi?
In Eisai’s 18-month study of nearly 1,800 people, Leqembi appeared to delay early-stage patients from getting worse by about five months.
The study measured people on an 18-point scale that follows their mental and functional abilities. People who got the drug still worsened but not as quickly as those given an inactive substance. The difference was nearly half a point on that scale by the study’s end.
Experts are divided over how meaningful the difference is.
Dr. Matthew Schrag is a neurology researcher at Vanderbilt University in Tennessee. He said, “Most patients won’t notice the difference.”
Schrag and some other researchers believe a meaningful improvement would require at least a difference of one full point on the 18-point scale.
Others say slowing the disease early on - when people still function well - is important.
Hodes, the director of the National Institute of Aging, said, “Several months with better cognition, what’s that worth to you?”
He added, “I think there you can get a strong argument: If I can interact with my family, be independent for months ... that’s a very meaningful outcome.”
What are the risks?
Like other amyloid-targeting medications, Leqembi can cause brain swelling or small brain bleeds. In Eisai’s study, 13 percent of drug recipients had swelling, and 17 percent has small brain bleeds.
The likely reason for this is that amyloid plaques usually form around nerve cells in the brain but sometimes they get inside blood vessels, too.
Several Leqembi users died while taking the drug, including two people who were on blood-thinning medications. Eisai has said its Alzheimer’s drug did not cause their deaths.
But Gandy, the Alzheimer’s expert, said the greatest risk of serious bleeding would be among Leqembi users who take blood thinners. Older adults commonly take blood thinners to prevent or treat strokes.
Patients also may experience reactions from the drug that could include high body temperature, an upset stomach and changes in blood pressure.
Eisai says the drug should be available by January 23. A year’s worth of treatment is expected to cost $26,500. If insurance companies cover the drug, however, most people will not have to pay anywhere near that much.
Words in This Story
decline - n. the process of become worse in condition
landmark - n. a very important achievement
encourage - v. to make something more likely to happen
scale - n. a range of numbers that is used to show size, quality or strength
cognition - n. the activity of thinking, understanding, learning and remembering
interact - v. to talk or do things with other people
outcome - n. something that happens as a result of activity or progress
swelling - n. an area on someone body's that is larger than normal because of an illness or injury
plaque - n. a change in brain tissue that happens in Alzheimer's disease
stroke - n. a serious illness caused when a blood vessel in your brain suddenly breaks or is blocked